Success Stories
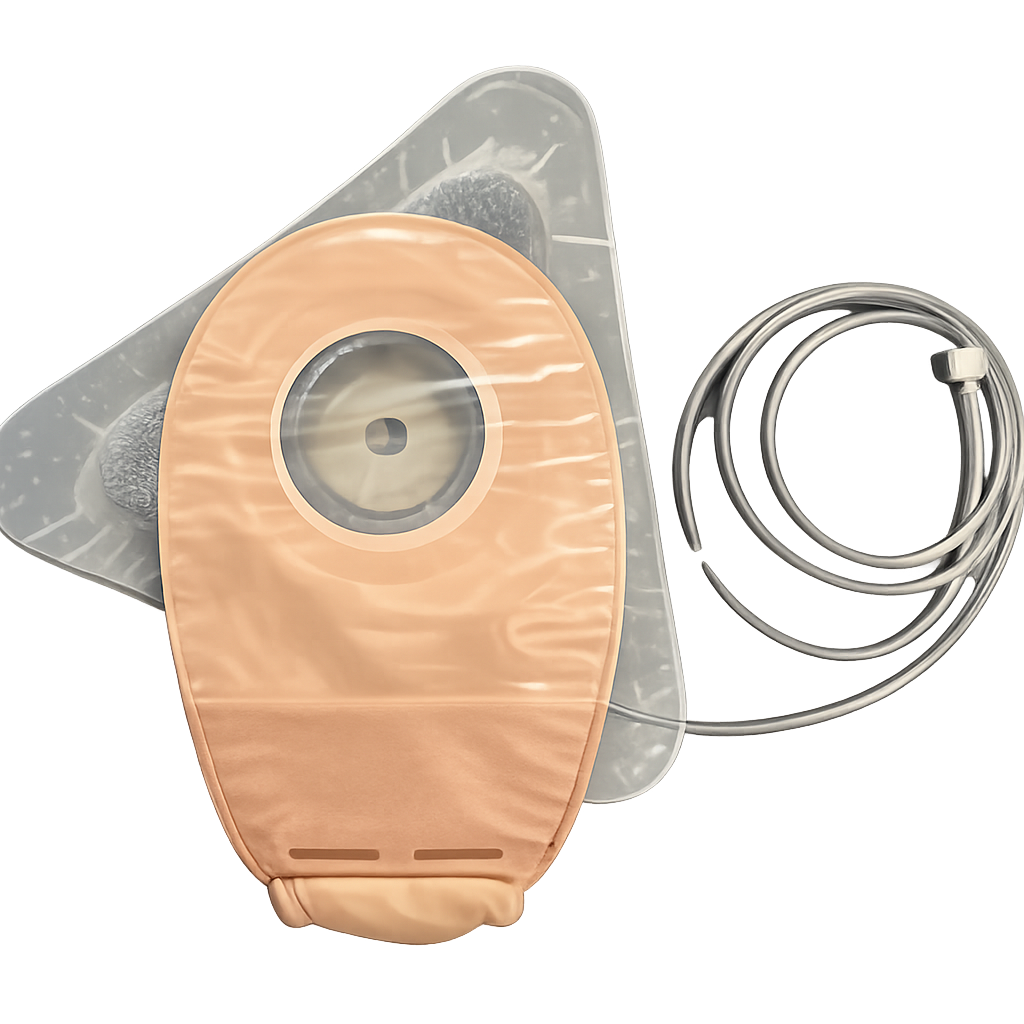
Fistula Solution
Class II
The PRoject:
Development of a single-use device intended for effluent management and promotion of healing of intact and injured skin surrounding enterocutaneous fistulae and ostomies.

Avio has been an amazing force multiplier to my company. Their team embodies collaboration and expertise, guiding us through the process of obtaining FDA Breakthrough Device Designation which was a major achievement. I’ve appreciated their support and insights through my product journey. I definitely recommend them to other entrepreneurs!”
Solutions
RA: Collaborated with internal team to develop regulatory strategy
QA: Generated Design History File and Risk Management File; Championed quality assurance best practices with internal team
R&D PM: Navigated internal team through product development lifecycle; V&V strategy, including Human Factors,Transit and Environmental Conditioning
results
- Secured Breakthrough Device designation
- Stood up a Quality Management System
- Created product development documentation processes
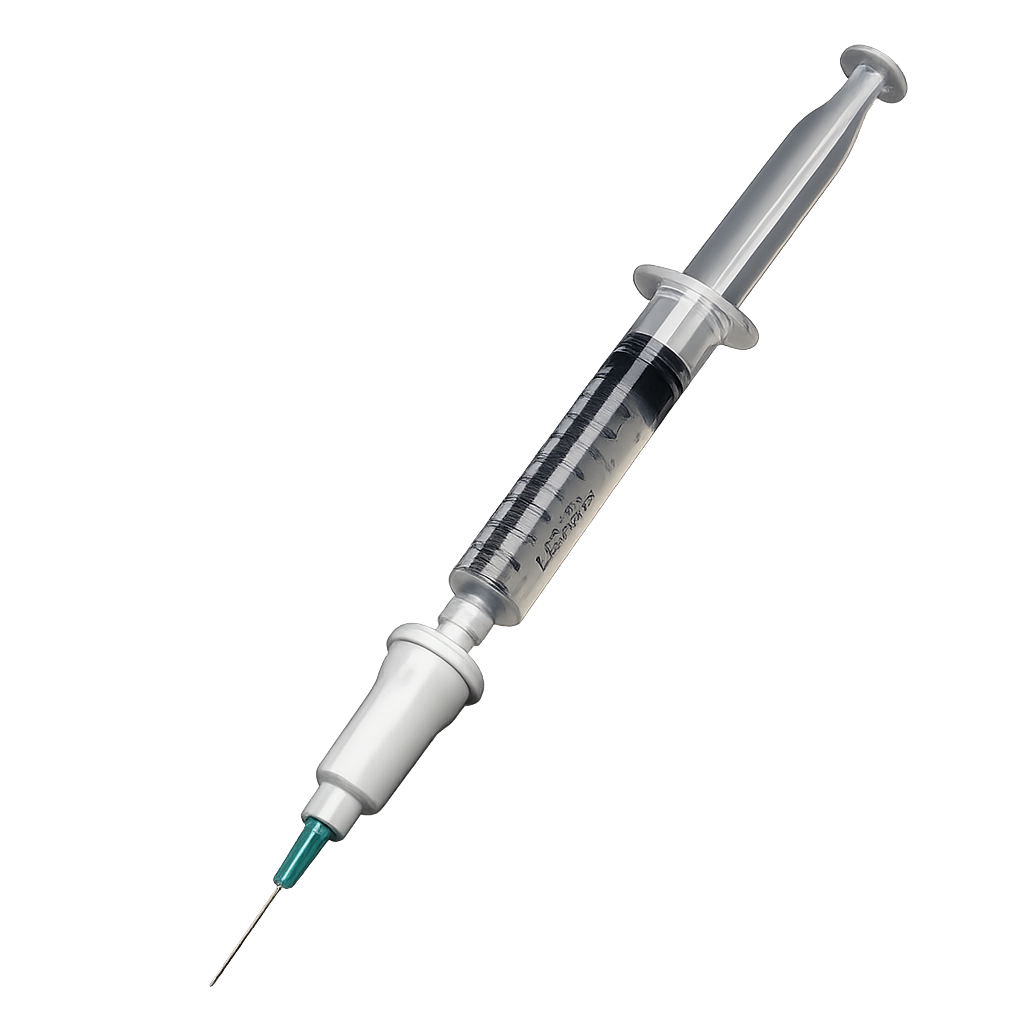
Agitated Solutions
Class II (Can & EU)
The PRoject:
Development of ultrasound contrast generating device.

The Avio team has been instrumental in Agitated Solutions’ success. They integrated seamlessly with my full-time team and have enabled me to reach our milestones more cost effectively. We have loved their positivity and expertise!”
Solutions
RA: Navigation of global regulatory landscapes and unique pharmaceutical-led combination product
QA: Led internal team through MDSAP audit in Canada and notified body audits in EU
R&D PM: Management of supply chain, contract manufacturing and testing vendors
results
- Secured medical license in Canada
- Management of application for CE marking and received CE approval
- Achieved MDSAP certification
- Two successful audits in the EU with zero findings